Not only optical detection
Most of the OPSIS monitoring instruments are based on optical techniques but there are exceptions. The concentration of oxygen in industrial processes can be measured optically but also with the help of a simple and robust electrochemical technique. The technology is based on the ceramic material zirconium dioxide and is perhaps best known as a "lambda probe" for controlling internal combustion engines.
HOW DOES THE ZIRCONIUM DIOXIDE TECHNOLOGY WORK?
Zirconium dioxide is a very durable ceramic consisting of crystallized, oxidized zirconium, ZrO2. In the basic version, a small block of zirconium dioxide is provided with two electrodes on opposite sides, this forms a "cell". One side with the electrode is exposed to a known oxygen content (reference), usually the 21 % of the ambient air, and the other side is exposed to the gas mixture which oxygen content is to be determined. When the cell is heated, the difference in oxygen content between the two sides gives rise to an electrochemically induced potential difference (voltage) between the electrodes. The voltage varies with the difference in oxygen content and by measuring the voltage the oxygen content in the gas mixture can be determined.

BROADBAND SENSOR
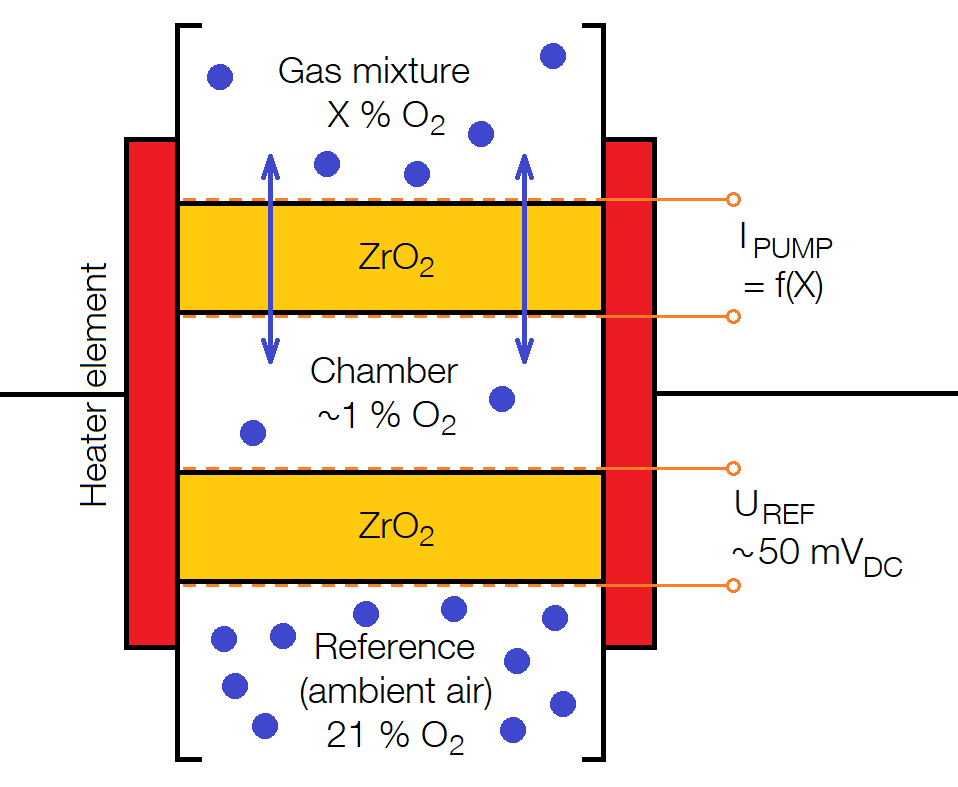
In the above design, the relationship between voltage and oxygen content is nonlinear which gives a rather poor measurement precision in certain content ranges. Nowadays, a so-called broadband sensor is therefore often used instead, which consists of two zirconium dioxide cells separated by a measurement chamber. As in the basic version, one side of the cell pack is then exposed to a known oxygen content and the other side to the gas mixture which oxygen content is to be determined. By driving a current through the cell closest to the gas mixture, you get an electrochemical pump that drives oxygen ions through the cell. In this way, the oxygen content in the measuring chamber can be regulated. The current and thus the pump power is controlled so that the potential difference across the second cell is kept constant at optimal sensitivity. The current through the first cell then becomes a measure of the oxygen content in the gas mixture. This provides a sensor with better precision than the original design.
BLOGS ABOUT OXYGEN MONITORING
Read more in our blog posts on oxygen monitoring and other articles on continuous emissions monitoring.
Operating Principle of an Oxygen Monitor

